The innovative company Continuity Biosciences and the international organization Breakthrough T1D have announced a collaboration within the latter’s Industry Discovery and Development Partnership (IDDP), with the goal of accelerating the clinical translation of the NICHE® biomedical platform for pancreatic islet transplantation in people living with type 1 diabetes (T1D).
Continuity Biosciences is a clinical-stage biotech start-up developing next-generation implantable platforms for the controlled and prolonged delivery of drugs, cellular reprogramming, and modulation of the immune system. Breakthrough T1D is a global organization, originally founded in 1970 in New York, dedicated to funding and accelerating research and translational initiatives to cure, prevent, and treat type 1 diabetes and its complications, also through strategic collaborations with industry partners.
The new collaboration will support a “phased” development pathway that builds on the preclinical results already achieved by Continuity Biosciences and will include both IND-enabling activities - that is, activities supporting regulatory readiness for clinical testing - and preparation for a first-in-human Phase 1/2a clinical study. In this context, Breakthrough T1D will support Continuity in the translational phase toward initial clinical evaluation, while maintaining a rigorous regulatory and manufacturing framework.
The goal is to make islet transplantation potentially accessible to a broader population of people with T1D; in preclinical models, the islet transplantation-based approach has shown the ability to restore normoglycemia.
The technology of the project
NICHE is a minimally invasive, refillable, implantable device placed subcutaneously for the transplantation of insulin-producing cells, intended for the treatment of patients with type 1 diabetes.
In more technical detail, NICHE is the only cell transplantation device that integrates a chamber designed to house insulin-producing cells (pancreatic islets) and a reservoir for the localized release of immunomodulatory agents directly at the graft site. In this way, immune modulation can be achieved at the graft level, reducing the need for systemic immunosuppression, which is often associated with side effects and limitations on patient eligibility.
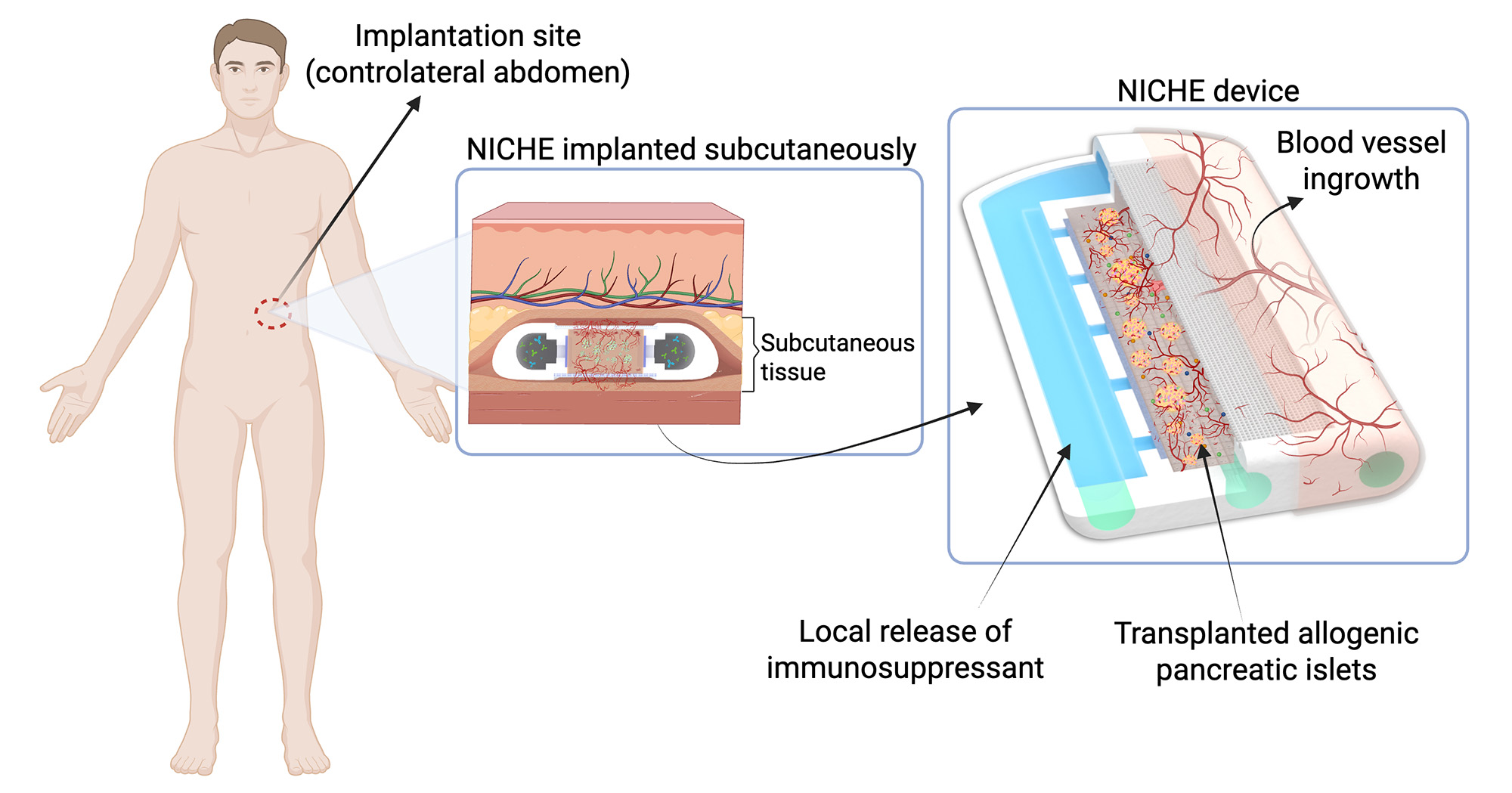
The planned Phase 1/2a “sentinel” study will assess, in adult patients with long-standing T1D, key aspects such as the safety of the device and the procedure, as well as the extent of vascularization and viability of the pancreatic islets.
The clinical study will involve some of the leading international experts in diabetes and pancreatic islet transplantation, thanks to collaboration with major clinical and academic centers in the United States. Continuity Biosciences will lead its development, regulatory aspects, manufacturing, and clinical execution in coordination with Breakthrough T1D.
The future of the venture
Alongside its U.S. headquarters, the company’s Italian presence in Turin - incubated since 2025 in I3P, the Innovative Companies Incubator of Politecnico di Torino - represents a strategic operational base for Continuity Biosciences, essential for coordinating research and development (R&D) activities and preclinical validation testing, while also contributing to the development of the platform and serving as a European hub for the company’s operations on the continent.
“Breakthrough T1D has solid experience in catalyzing significant progress in type 1 diabetes research. This collaboration will allow us to advance NICHE® along a rigorous clinical and regulatory pathway,” said Ramakrishna Venugopalan, Chief Executive Officer of Continuity Biosciences.
“We want to make this therapy potentially accessible to more people with type 1 diabetes by building a solid and scalable development pathway,” commented Marco Farina, General Manager Italy & Senior Director, Platform Development at Continuity Biosciences LLC. “This achievement stems from the work of a team spanning Turin and the United States and from the contribution of engineers trained at the Politecnico di Torino. With the support of I3P, we are strengthening Turin as a strategic European hub for the development and clinical validation of our technologies.”

